The U.S. Government Accountability Office (GAO) publishes independent audits at the request of consumer advocates, watchdog groups, and members of the United States Congress. One of the most recent publications involved the U.S. Food and Drug Administration (FDA) post market surveillance process, including an analysis of management & oversight across agency functions and employees.
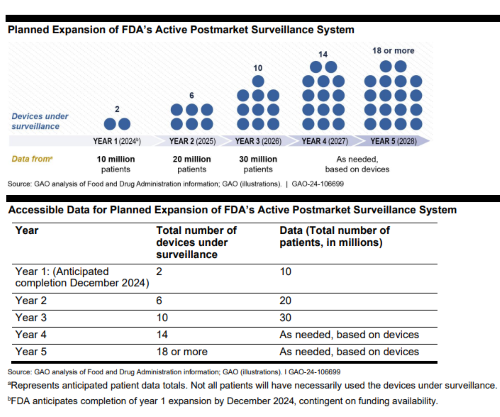
Additionally, the post marketing study uncovered hurdles FDA encountered during the medical device audit. The final 27-page report included feedback on whether the agency was efficient while handling problems and protecting data integrity. These details offer a snapshot of how a regulatory body operates in real time across the life science industry, and where efficiency could be examined.
What is a Post Market Surveillance System?
The U.S. Food and Drug Administration (FDA) has an approved regulatory framework & database (MAUDE) designed to connect consumer complaints and compliance adverse events to a product code and medical device manufacturer. This post market surveillance system (PMS) is a validated FDA regulatory process designed to increase transparency and continuous improvement.
Adverse Events
A post market surveillance system is the market listening mechanism between real world evidence and analysis. Software platforms are used as a global harmonization resource for monitoring adverse events (e.g. poisoning, adverse reaction) across countries, devices & the cycle of patient life.
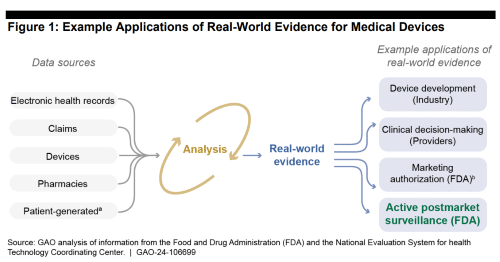
Additionally, the post market surveillance system is designed to help collect HIPAA-compliant patient data & medical device product ID info in a single source of postmarket truth. Patent-generated evidence (e.g. FitBit, Apple Watch) is a growing data source HCPs recognize at the point of care which medical device manufacturers could use during product design & commercialization.
Patient Safety
Additionally, the post marketing surveillance process uses incoming data to search for patient safety trends proactively. The future may include post marketing requirements that help educate the population’s health, and when label changes should be communicated to consumers. Finally, when a negative safety trend begins to accelerate, FDA may consider if the marketing approval of the product should be removed.
What is a Unique Device Identifier (UDI)?
The unique device identification (UDI) is an exclusive code related to a specific medical device. Each UDI promotes a transparent identification system that helps life science employees trace a product using a numerical & alphabetical format on the device label. Each unique device identification number includes a mix of production and device identifier (DI) data based on the manufacturing process. Further, the elements of the production identifier (PI) portion of the UDI code include this information on each device label:
- Manufacturing date
- Manufacturing batch number (or lot number)
- Device serial number
- Device expiration date
- Distinct identification code (for combination products)
FDA mandates that any patient documentation needed to uncover the root cause of a medical device’s problem be included in the safety profile risk analysis. Ideally, the risk analysis would also contain information from a patient’s EHR health records and/or the payer’s claim data or pharmacy data. Both of these are widely considered reliable post market surveillance data sources.
A full transcript and highlights of the GAO report can be found here.
About RCA’s Medical Device Consulting Services
The regulatory compliance process surrounding the medical device industry involves strict adherence to pre/post market information throughout a device’s life cycle. Even a single compliance issue you have can turn into a significant effect on your business. Regulatory Compliance Associates can help guide you through any stage of the medical device consulting process, with capabilities during product development through the regulatory clearance/approval of your product.
Our team of over 500 medical device consulting Experts — including former FDA officials and regulatory compliance leaders in the field of medical device regulation — will work with your company to create a quality assurance and regulatory compliance approach tailored to your products and regulatory needs. Regulatory Compliance Associates works with international Fortune 100 companies, venture capital start ups, and companies of all sizes and shapes. Our regulatory compliance solutions for law firms include remediation for warning letters, FDA 483s, import bans, or consent decrees. Very few regulatory compliance services have the same regulatory compliance expertise in a variety of medical fields.
Medical Device Cybersecurity
For medical device manufacturers, technology can be a double-edged sword. The innovative technologies that elevate the quality of life for patients can also be used to potentially undermine the organization using the device. The consequences can affect the device itself if Regulatory Compliance Associates medtech consultants do not implement good IoT cybersecurity and FDA cybersecurity protocols.
At Regulatory Compliance Associates, we offer a wide variety of medical device cybersecurity services to ensure your product is protected from cyberattacks. With a well-planned design, along with full visibility of product development and the supply chain, Regulatory Compliance Associates medical device consulting Experts can help strengthen your device’s cybersecurity. We partner with medical device companies in each phase of the design cycle. The RCA medical device consultant team understands protecting inputs from threat exposure and hardening outputs for regulatory compliance & FDA submission approval.
- SaMD Consulting
- Threat Modeling
- Proof of Concept
- Quality Assurance Services
- TIR 57 & TIR 97
- ISO 62304
- ISO 27001
Regulatory Affairs
Regulatory affairs is Regulatory Compliance Associates® backbone. We handle more regulatory consulting submissions in a month than many manufacturers do in a lifetime. Our regulatory compliance consulting Experts have experience working with the FDA, global regulatory bodies and/or agencies, and notified bodies worldwide. Therefore, you can count on us for in-depth and up-to-date insights that increase speed-to-market.
As a trusted regulatory affairs consultant, our FDA veterans and regulatory consulting team represents Regulatory Compliance Associates® as one of the top medical device consulting firms. We’ll help you navigate the medical device consulting difficulties associated with new product submissions. Regulatory Compliance Associates® medical device consulting company has expertise in both the approval process and post-approval support.
- New Product Approval
- Post-Approval Support
- Outsourced Staffing
- EU MDR
- Combination Products
Compliance Assurance
Increasingly, life science companies are feeling the pressure of greater scrutiny by regulators, and responding by developing sustainable compliance strategies. Whether it’s preparing for an audit, developing a response to an FDA finding, or remediation to an adverse event, Regulatory Compliance Associates® can help.
Our network of over 500 compliance consulting Experts are industry professionals & FDA, MHRA & EMA veterans. This allows Regulatory Compliance Associates® to handle both simple and complex regulatory compliance challenges within medical device consulting scenarios.
- Gap Assessments
- Internal Audits
- Employee Training
- Notified Body Response
- Data Integrity
Quality Assurance
Regulatory Compliance Associates® is globally known for quality assurance services, including quality system assessments, strategy, implementations, and quality metrics to ensure continuous improvement. Each Regulatory Compliance Associates® medical device consultant is a quality expert with experience spanning major corporations and start-ups. RCA’s quality assurance services experts know firsthand how to achieve, maintain, and improve quality, and we excel in transferring this knowledge to your organization.
In the medical devices field, quality assurance (QA) is more than merely ensuring the quality of a finished product. You need the tools to monitor and regulate every process from the design of a new product to continued quality compliance as the device is sent to market. At Regulatory Compliance Associates®, we offer you the quality assurance services you need to monitor these processes and ensure quality compliance every step of the way.
With more than 20 years of experience working with medical device consulting companies, Regulatory Compliance Associates® trusted medical device quality assurance consultant team is fully equipped to handle your unique QA needs.
- ISO13485
- 21 CFR 210
- 21 CFR 211
- Outsourced Staffing
- MDSAP
- Facility Validation
- Equipment Validation
- Quality Metrics
Remediation Services
Regulatory Compliance Associates® is widely recognized within medical device consulting companies & the life science industry for our remediation services & support. Regulatory Compliance Associates® ability to help companies successfully resolve complex regulatory challenges has a proven track record of success. Our medical device consulting services include significant experience with the development of responses to 483 Observations, Warning Letters, Untitled Letters, and Consent Decrees.
- Regulatory Action
- Regulatory Compliance
- Regulatory Enforcement
- Warning Letter
- 483 Observation
- Oversight Services
Our value goes beyond the initial response by helping companies successfully execute their action plans, develop an improved compliance culture tailored to the needs of their business, and ultimately move beyond the regulatory action to emerge as a stronger business. We negotiate demands of remediation services with insight and the clear advantage of our medical device consulting expertise. Our deep industry experience makes partnering with Regulatory Compliance Associates® a competitive differentiator in the remediation services space.
- Quality System
- Technical File
- Design History File
- Data Integrity
- cGMP
Strategic Consulting
Whether it’s a strategy, a technical plan, or a project, Regulatory Compliance Associates® medical device consultancy can help ensure a successful project. Regulatory Compliance Associates® due diligence consulting can deliver your project on time, and on budget, and you’re never embroiled in a costly mistake.
Our medical device consulting Experts are industry Experts are here to provide the unique insight you need before an M&A deal, through a staffing crisis and in every area of your product’s development and life cycle. As the trusted medical device manufacturing consultants of thousands of companies around the world, we have the knowledge and expertise needed to deliver exceptional results to your business — no matter your size or unique needs.
- Manufacturing Optimization
- Product Lifecycle Management
- Mergers & Acquisitions (M&A)
- Due Diligence
- Device Vigilance
- Risk Management Plan
- Product Complaints
- Medical Information
About Regulatory Compliance Associates

Regulatory Compliance Associates® (RCA) provides medical device consulting to the following industries for resolution of life science challenges:
We understand the complexities of running a life science business and possess areas of expertise that include every facet of R&D, operations, regulatory affairs, quality, and manufacturing. We are used to working on the front lines and thriving in the scrutiny of FDA, Health Canada, MHRA and globally-regulated companies.
As your partners, Regulatory Compliance Associates can negotiate the potential minefield of regulatory compliance and regulatory due diligence with insight, hindsight, and the clear advantage of our unique expertise and experience.
- Founded in 2000
- Headquartered in Wisconsin (USA)
- Expertise backed by over 500 industry subject matter experts
- Acquired by Sotera Health in 2021
About Sotera Health
The name Sotera Health was inspired by Soteria, the Greek goddess of safety, and reflects the Company’s unwavering commitment to its mission, Safeguarding Global Health®.
Sotera Health Company, along with its three best-in-class businesses – Sterigenics®, Nordion® and Nelson Labs®, is a leading global provider of mission-critical end-to-end sterilization solutions and lab testing and advisory services for the healthcare industry. With a combined tenure across our businesses of nearly 200 years and our industry-recognized scientific and technological expertise, we help to ensure the safety of over 190 million patients and healthcare practitioners around the world every year.
We are a trusted partner to 5,000+ customers in over 50 countries, including 40 of the top 50 medical device companies and 9 of the top 10 pharmaceutical companies.
Commitment to Quality
Our Certificate of Registration demonstrates that our Quality Management System meets the requirements of ISO 9001:2015, an internationally recognized standard of quality.
To begin the Regulatory Compliance Associates® scoping process today, please enter your information in the blue form below and click the submit button at the bottom of the webpage.